-
01
Industrial Battery
The Best Lithium Battery Solution and Product
Trustworthy Battery Provider and Partner -
02
E-bike Battery.
A vital part of any E-bike
The best selection of all shapes and types for you -
03
ESS Battery
Custom energy storage battery ISO9001:2015
quality management system manufacturer -
04
LiFePO4 Battery
10+ year's manufacturer
Maximum Relability & Maximum Safety

New Products
-
Bike
brands -
Special
offers -
Satisfied
clients -
Partners throughout
the USA
WHY CHOOSE SOSLLI
-
why choose lithium-ion battery
Lithium-ion battery chemistry has become the industrial battery chemistry of choice for everything from electric vehicles to energy storage, from commercial to utility to military applications. The advantages of lithium-ion battery chemistries include.
· Higher energy density delivering long run times between charges
· Compact size and light weight
· Faster recharge times
· Wider operating and storage temperature ranges
· Long cycle life
· Maintenance free operation -
SOSLLI Services
· 7/24 service, Quick response to clients' inquiries within 12hrs
· Exclusive and unique solution provided to our clients by our experienced and professional engineers/researchers.
· Solve the problems promptly (24hrs) if anything wrong with our products' quality.
· The leading lithium battery manufacturer acquired ISO9001: 2015, UL, CE, RoHS, UN38.3, MSDS etc. certifications.
· All SOSLLI battery products cover the US$1,000,000 products liability insurance by CPIC. -
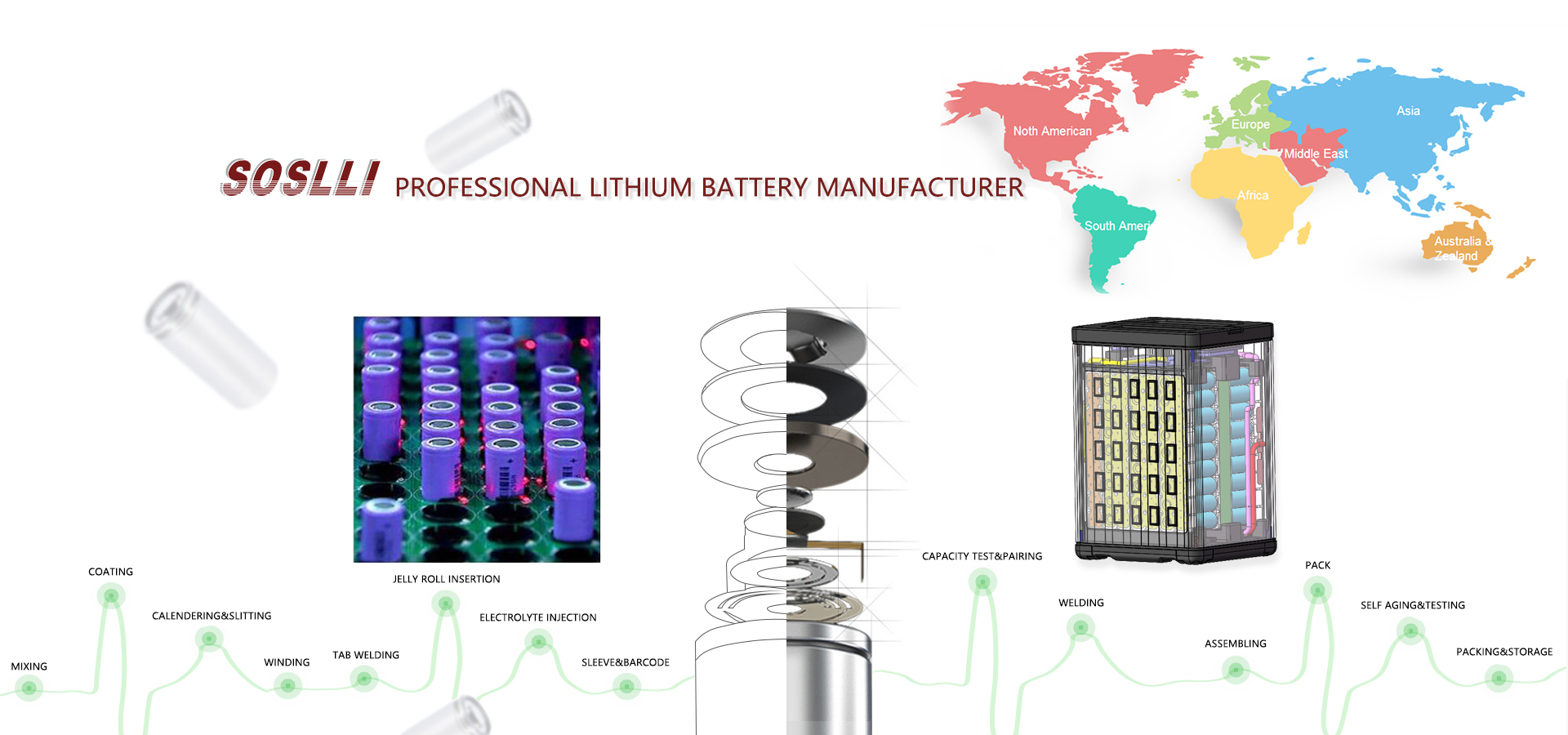
SOSLLI Advantages: (Strictly Implement ISO9001 Quality System)
1. Production System 1600 employees, High quality equipments imported from Japan and South Korea creates an automatic production line.
2. Production Quality 4-Dimensional quality system, 110 QC staff, 100% inspection. The industry top 10 suppliers, battery acquired CE, RoHS, UL, KC, IEC62133, MSDS,UN38.3, etc.
3. OEM/ODM (Flexibly Customizable) 10+ years experience, Provide customers with the tailored battery module to meet different application requirements.
4. Continuous Innovation 66+ professional engineers, R&D center and testing center with rigorous team organization, advanced equipments and enormous investment.
-


Corporate spirit
Sincere communication, commit to the achievement of customers and employees. Team work and focus on supply the best Li-ion battery products and services.
-


Corporate value
Honest and diligent, Innovative and efficient,Quality and service, Cooperation and multi-win.
-


Corporate mission
Become a leading technology, excellent quality, innovative and efficient, and first-class service international new energy enterprise!